Offers three-week, closed-system TIL manufacturing ensuring precision, scalability, and reliable GMP-grade cell therapy production
uBriGene Biosciences is a global cell and gene therapy CDMO with proven expertise in complex tumor-infiltrating lymphocyte (TIL) manufacturing processes that require extensive open manipulations.
Optional caption section: This caption text can be used under any image or content block as-needed. Use cases are figure legends, citations, image descriptions
Our proprietary automated platform delivers consistent, scalable, and potent TIL products within three weeks, advancing your immunotherapy development with confidence.
These advantages not only improve the efficiency and reliability of the manufacturing process but also ensure a more cost-effective production of cells.
Consult with us for seamless IND submissions, regulatory compliance, and cutting-edge technology in CAR-NK and CAR-T CDMO manufacturing services.
Activated DMF on file with the FDA

Optional caption section: This caption text can be used under any image or content block as-needed. Use cases are figure legends, citations, image descriptions
We provide rapid/comprehensive QC release testing, including viability, potency, purity, and safety.
Yield & Viability
TILs derived from both lung and melanoma tumor samples exhibited consistently high cell yield and viability throughout the manufacturing process.
Purity
Expanded TILs showed a consistently high proportion of CD4⁺ and CD8⁺ subsets, with minimal monocyte and NK cell contamination.
Potency
Potency and IFN-γ release assays confirmed strong TIL functionality, highlighting a robust, reproducible, and scalable expansion process suitable for clinical manufacturing.
As an experienced cell and gene therapy CDMO, uBriGene addresses key pain points in TIL manufacturing process simplification and automation.
We have successfully expanded TILs ex vivo to clinical doses, incorporating: G-Rex, Wave 25 Rocker Bioreactor, Sepax C-Pro Cell processing System, and Lovo cell processing system.
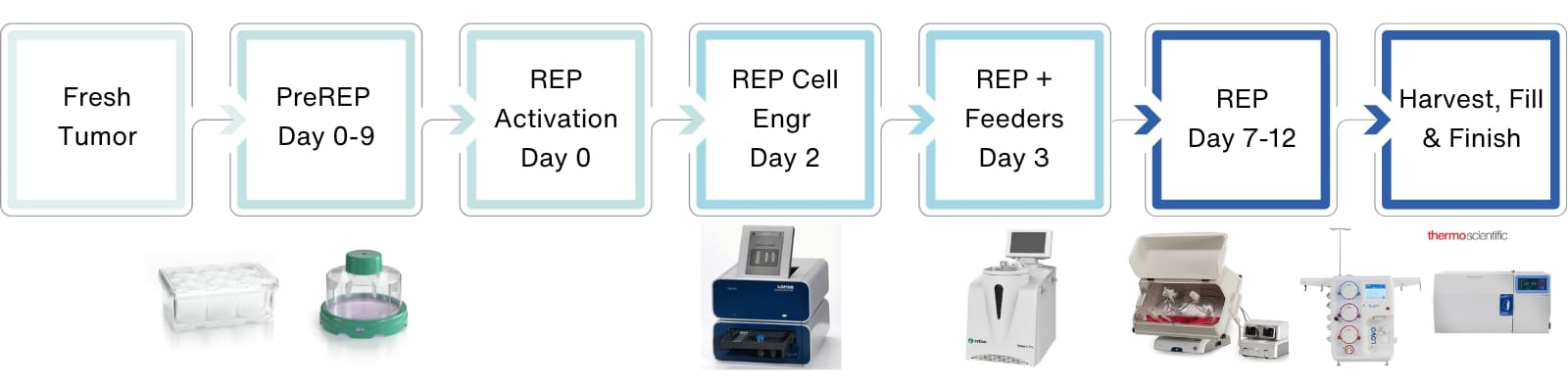
Figure 1. Schematic representation of the preREP and REP TIL manufacturing process
Request a quoteOur quality control testing includes rigorous testing for cell identity, purity, potency, and safety. Additionally, our regulatory affairs team can support IND filing.
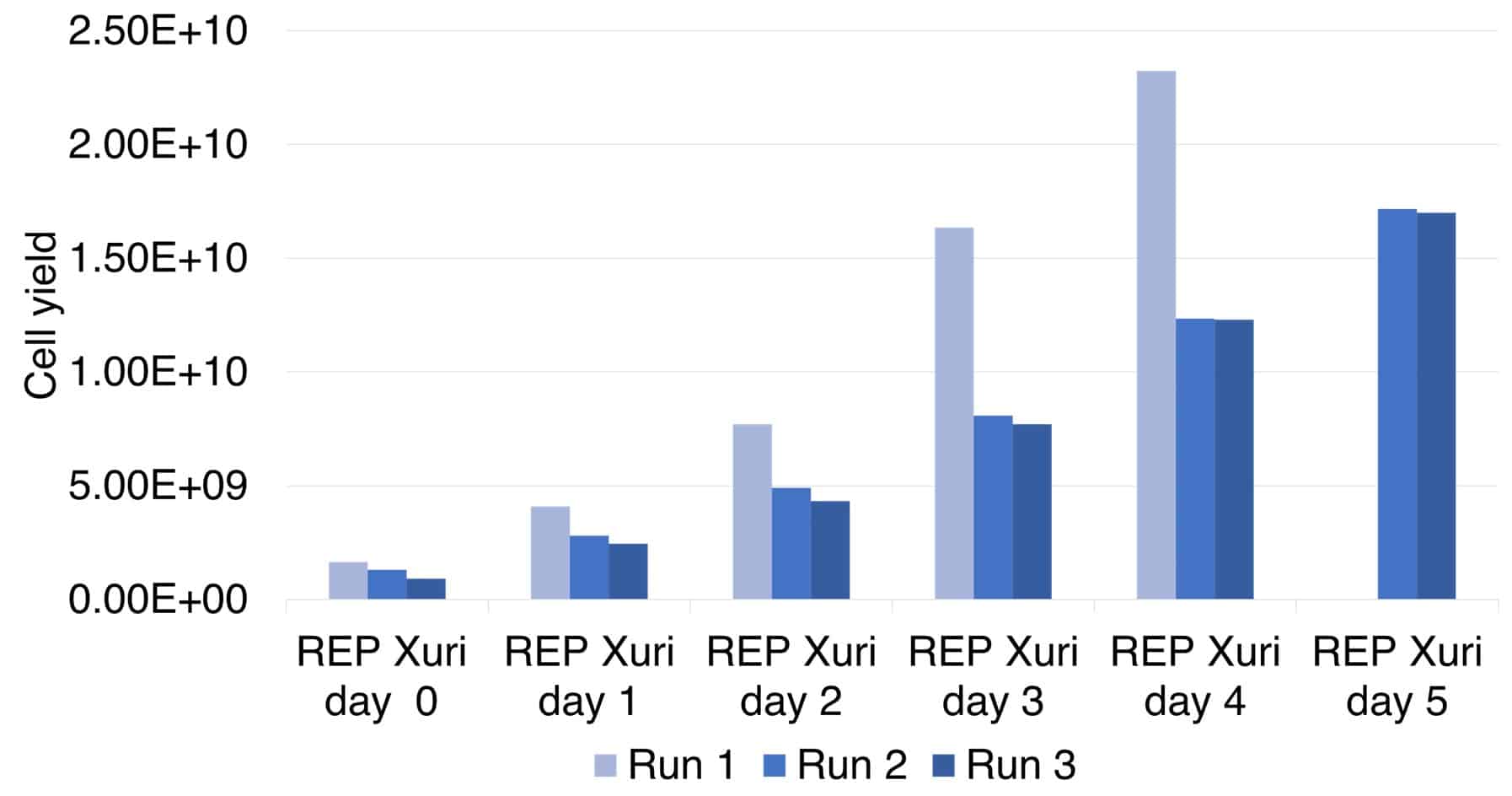
Figure 2. Growth kinetics of TILs during expansion in the Xuri Cell Expansion System
Request Project Scope & CostThe significance of our typical MSC (Mesenchymal Stem Cell) production process workflow lies in its integrated, one-stop comprehensive service, ensuring efficiency, consistency, and quality at every stage of GMP stem cell manufacturing. This includes tissue sample collection, stem cell separation and amplification, cell bank construction, MCB verification, large-scale stem cell production, harvest and aseptic filling, followed by quality inspection and product release.

MSC GMP manufacturing workflow diagram
Request a quoteAt uBriGene, we uphold the highest standards of quality control and assurance throughout our GMP iPSC/MSC manufacturing process, ensuring that every product meets stringent criteria for safety, purity, and efficacy.
| Test Item | Assay |
|---|---|
| Sterility | Immersion sterility tests(B/F) |
| Mycoplasma Testing | Culture and cell indication assay |
| Endotoxin | Kinetic chromogenic LAL |
| Cell Viability | Trypan blue |
| Pluripotency Characteristics | Flow cytometry |
| Embryoid body formation | |
| Genetic stability | STR genotyping |
| Karyotype analysis | |
| Viral factor inspection | Transmission electron microscopy (TEM) |
| In vitro assay for adventitious virus contaminant | |
| Fluorescent Product Enhanced Reverse Transcriptase (FPERT) method |
*Outsourced testing
Stem cell production undergoes rigorous testing using advanced techniques to ensure regulatory compliance and exceed customer expectations. Testing is conducted at every stage, from raw material sourcing to final GMP iPSC release, including but not limited to the following tests:
Identity and Purity Testing: Ensuring the genetic integrity and purity of our iPSC lines.
Sterility Testing: Confirming the absence of microbial contaminants.
Viability and Proliferation Assays: Assessing the health and proliferative capacity of our iPSCs/MSCs.
Genetic Stability Assessment: Monitoring for genetic alterations during expansion and passaging.
The advantages of membrane matrix in downstream chromatography for plasmid purification include requiring only 30% of the time compared to conventional resin. Therefore, plasmid DNA is manufactured faster and at a much lower cost.
At uBriGene, we uphold the highest standards of quality control and assurance throughout our GMP iPSC/MSC manufacturing process, ensuring that every product meets stringent criteria for safety, purity, and efficacy.

Learn how to accelerate TIL Therapy with proven manufacturing expertise
Unveil the current manufacturing approaches for LVV and its deployment in cell therapies.
Unlock safer regenerative therapies with Genome-Safe iPSC RPM for clinical success.
Discover AAV manufacturing capabilities, scalable processes, and testing platform.
We provide comprehensive GMP manufacturing services, including donor screening, iPSC generation, MSC production, cell banking, process development, quality control, and regulatory support. Our services also include analytical method development and GMP-grade production for clinical applications.
uBriGene uses an efficient mRNA-LNP reprogramming mix for iPSC generation, available in both research and GMP grades. We also offer GMP-grade Cas9 nuclease, Cas9 mRNA, and sgRNA production for gene editing, as well as single-stranded DNA and linear closed-end dsDNA manufacturing. Yes, we offer full CRISPR gene editing services.
Mesenchymal stem cells (MSCs) are stromal cells used in treatments like myocardial infarction, fibrosis, and inflammatory diseases. MSCs, derived from adult and neonatal tissues, are valued for tissue regeneration, immune modulation, and low immunogenicity.
Yes, we offer customizable production services, including donor cell selection, protocol optimization, and comprehensive cell banking (MCB, WCB) under GMP conditions.
We conduct rigorous testing for cell identity, purity, potency, and sterility, along with karyotyping and genetic stability checks. Our processes comply with FDA, EMA, NMPA, and other regulatory guidelines.
Turnaround times vary by project scale—iPSC production takes weeks to months, while MSC production takes several weeks. We also offer regulatory support for clinical trial submissions.
We have extensive experience in large-scale production, using bioreactors and automation technologies. Our QA processes include SOPs, batch record documentation, and regular audits to ensure reproducibility and consistency.
We provide comprehensive documentation, including batch records and certificates of analysis, for regulatory submissions.
We employ stringent quality control measures, including regular monitoring of cell cultures for genetic stability using karyotyping, SNP analysis, and other techniques to detect any abnormalities.
Yes, we offer iPSC differentiation services to generate specific cell types, such as cardiomyocytes, neurons, or hepatocytes, for research or drug discovery purposes.
We follow strict quality assurance processes, including SOPs (Standard Operating Procedures), batch record documentation, and regular audits, to ensure reproducibility and consistency in cell manufacturing.
Yes, we have extensive experience working with regulatory agencies to support cell therapy approvals and regulatory submissions, including the FDA, EMA, NMPA, and TGA.
Take the next step in your CAR journey. Discuss your project requirements and see how our CAR-NK & CAR-T CDMO services can support your goals.
Simplified & Automated TIL Manufacturing processes
Integrated End-to-End Services
Experienced TIL Experts
Global Cell and Gene CDMO Footprint
Sit aliquam elit vitae bibendum lectus blandit dictum. Nisi porta elementum sed diam eleifend pellentesque. Quis aenean quam.
Request now
Optional caption section: This caption text can be used under any image or content block as-needed. Use cases are figure legends, citations, image descriptions
Ask the plasmid DNA expert! With over 300 GMP batches in our track record and our cost-effective platform technology, we can help accelerate your clinical programs.