uVivo™ LVV is a targeted in vivo CAR-T platform that enables precise CAR delivery to T cells and rapid CAR-T generation in vivo. Single-dose administration supports low cost, off-the-shelf cancer therapies.

The uVivo™ LVV platform enables specific and rapid in vivo generation of chimeric antigen receptor (CAR)-T cells. Precise T-cell delivery is achieved through dual engineering: detargeted uVivo-CocalG and CD7 nanobody–mediated retargeting.
In vivo CAR-T generation (no ex vivo processing required
Detargeted uVivo-CocalG eliminates native broad tropism
T-cell targeting enabled by a CD7 nanobody with an optimized membrane anchoring sequence
High specificity with minimal off-target transduction
Validated efficacy in vivo
High productivity driven by optimized Ubri-CocalG and CD7 T-cell binder
The various Rep/Cap plasmids and helper plasmids have been tested for high quality and high yield in GMP AAV production.
We have assisted our clients in obtaining IND clearance with AAV produced using these AAV helper plasmids.
| Catalog# | Description | Particles per Vial | Product Grade | Price |
|---|---|---|---|---|
| HU-IPSC-EXOSOME | High-quality exosomes / Extracellular vesicles produced from Human iPSC master cell bank, purified via chromatography. | 20 billion particles | Research | |
| Varies | GMP | Please Inquire | ||
| HU-MSC-UC-EXOSOME | High-quality exosomes produced from the Human umbilical cord MSC bank, purified via chromatography. | 20 billion particles | Research | |
| Varies | GMP |
Note: This product is intended for in vitro research and/or manufacturing use only. Biological samples and any materials derived from these samples are for in vitro research purposes only.
Request a quotePreclinical studies demonstrate highly specific T-cell targeting, with minimal to no transduction in non-target tissues
uVivo™ LVV is powered by detargeted Ubri-CocalG and CD7 nanobody–mediated T-cell targeting
Sit aliquam elit vitae bibendum lectus blandit dictum. Nisi porta elementum sed diam eleifend pellentesque. Quis aenean quam.
Schedule a Discussion

Optional caption section: This caption text can be used under any image or content block as-needed. Use cases are figure legends, citations, image descriptions
LVV Turbo™ 293TH suspension cells, 5x higher productivity
Fully closed downstream, eliminating the final sterile filtration, up to 70% recovery
The uVivo™ LVV platform is an in vivo CAR-T technology that enables precise delivery of CAR payloads to T cells, allowing rapid generation of CAR-T cells in vivo.
uVivo™ LVV particles are third-generation, self-inactivating, replication-deficient lentiviral vectors pseudotyped with a detargeted fusogen (uVivo-CocalG) and equipped with a CD7 T-cell binder for targeted delivery.
When combined with the high-yield, scalable LVV Turbo™ manufacturing platform, the off-the-shelf uVivo™ LVV platform has the potential to significantly improve the accessibility and affordability of CAR-T therapies.
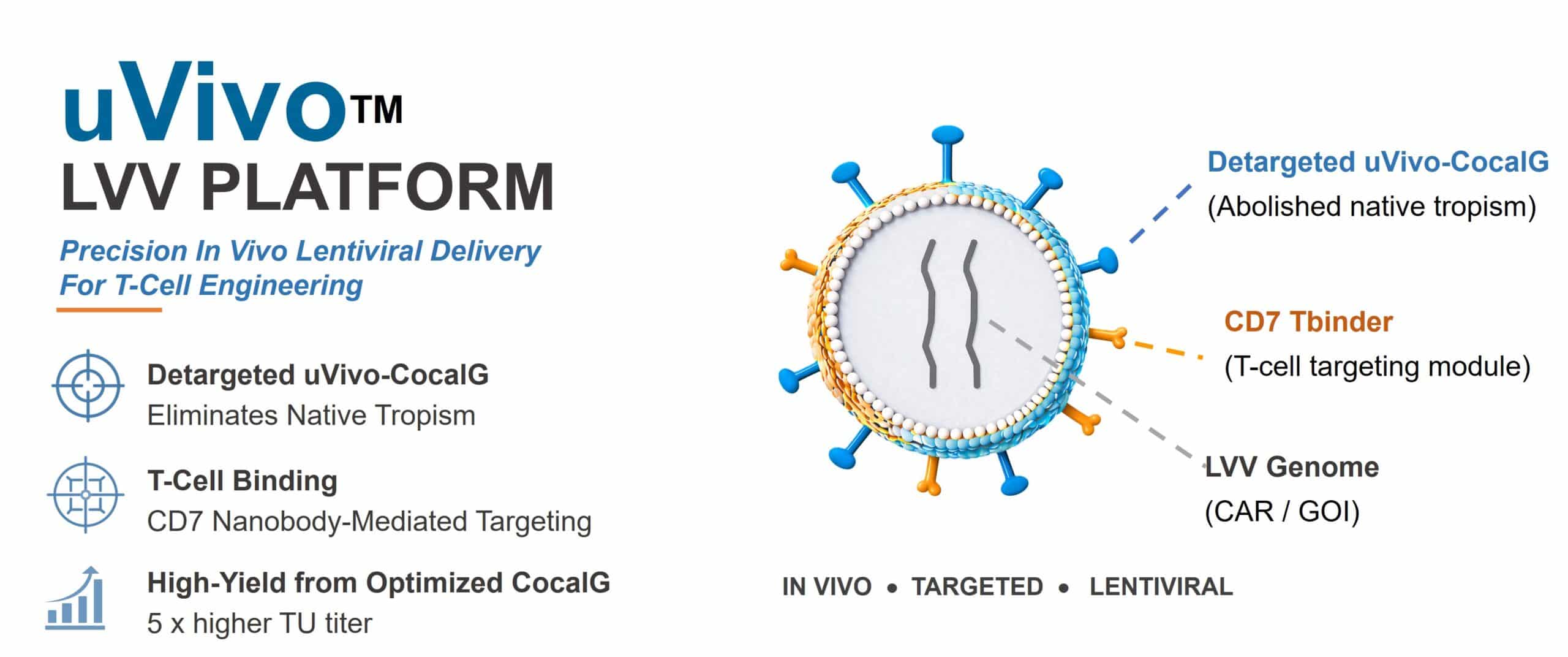
Fig. 1. uVivo™ LVV platform schematic illustrating targeted in vivo CAR-T delivery.
The uVivo™ platform uses a multi-step engineering strategy to achieve precise T-cell tropism. A sequence-optimized, detargeted Ubri-CocalG eliminates native receptor interactions while retaining fusion activity, and a CD7 nanobody–based binder enables targeted T-cell delivery, with transmembrane optimization increasing LVV activity by up to 10-fold.
This detargeting and retargeting approach enables selective in vivo T-cell transduction while minimizing off-target delivery to other tissues.
Engineered CocalG enables detargeted and retargeted LVV delivery, making it more suitable than VSV-G for targeted in vivo CAR-T applications. Schedule a discussion.
Using the uVivo™ LVV platform, transduction titers for CAR payloads (measured by flow cytometry in PBMCs) can reach 3E9–2E10 TU from a 10 L batch, enabling the production of thousands of in vivo CAR-T doses.
This high productivity is driven by several key innovations:
Sequence-optimized Ubri-CocalG delivers ~3× higher productivity compared to the wild-type sequence
Optimized Ubri-CD7 T-cell binder (with engineered transmembrane domain) achieves up to a 10-fold increase in transduction efficiency
GOI sequence optimization further enhances TU titers by up to 5×
Integration with the LVV Turbo™ manufacturing platform, featuring a high-yield suspension cell line and a closed downstream process for scalable production.
The data below illustrates the detargeting (uVivo-CocalG), retargeting via CD7 T-cell binder optimization, and the transduction titer of our uVivoTM LVV platform.
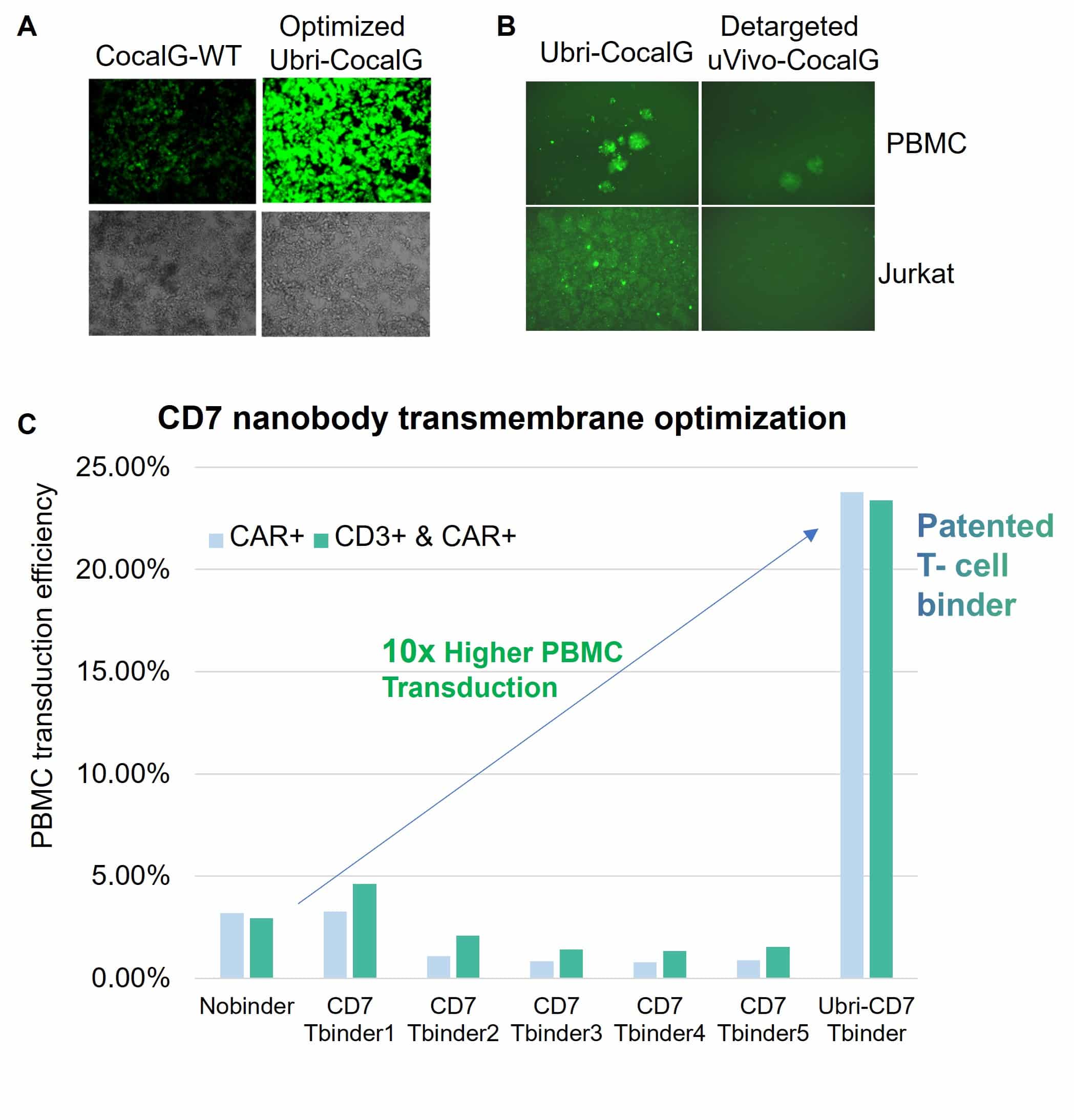
Fig. 2. uVivo-CocalG engineering for high yield and T-cell targeting.
A. Ubri-CocalG increases productivity. Sequence-optimized Ubri-CocalG shows ~3× higher gene expression than the wild type in 293T cells (24 h post-transduction).
B. Detargeted uVivo-CocalG eliminates native tropism. GFP LVVs pseudotyped with Ubri-CocalG or uVivo-CocalG were used to transduce PBMCs or Jurkat cells; images were captured 24 h post-transduction.
C. Optimized CD7 T-cell binder enhances transduction. Transmembrane variants were screened in PBMCs using uVivo-CocalG–pseudotyped LVVs. The optimized binder achieved ~10× higher transduction efficiency; CAR⁺ (blue) and CD3⁺CAR⁺ (green) cells were measured by flow cytometry.
In preclinical studies, humanized mouse models of multiple myeloma treated with a single intravenous dose of anti-BCMA uVivo™ LVV particles showed complete tumor clearance.
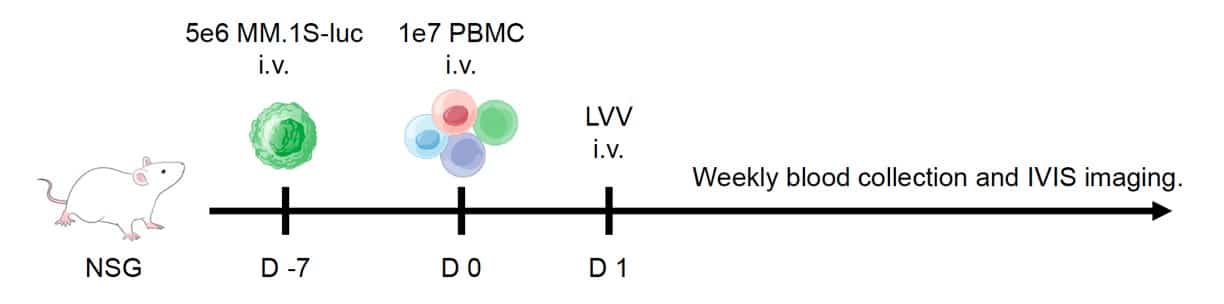
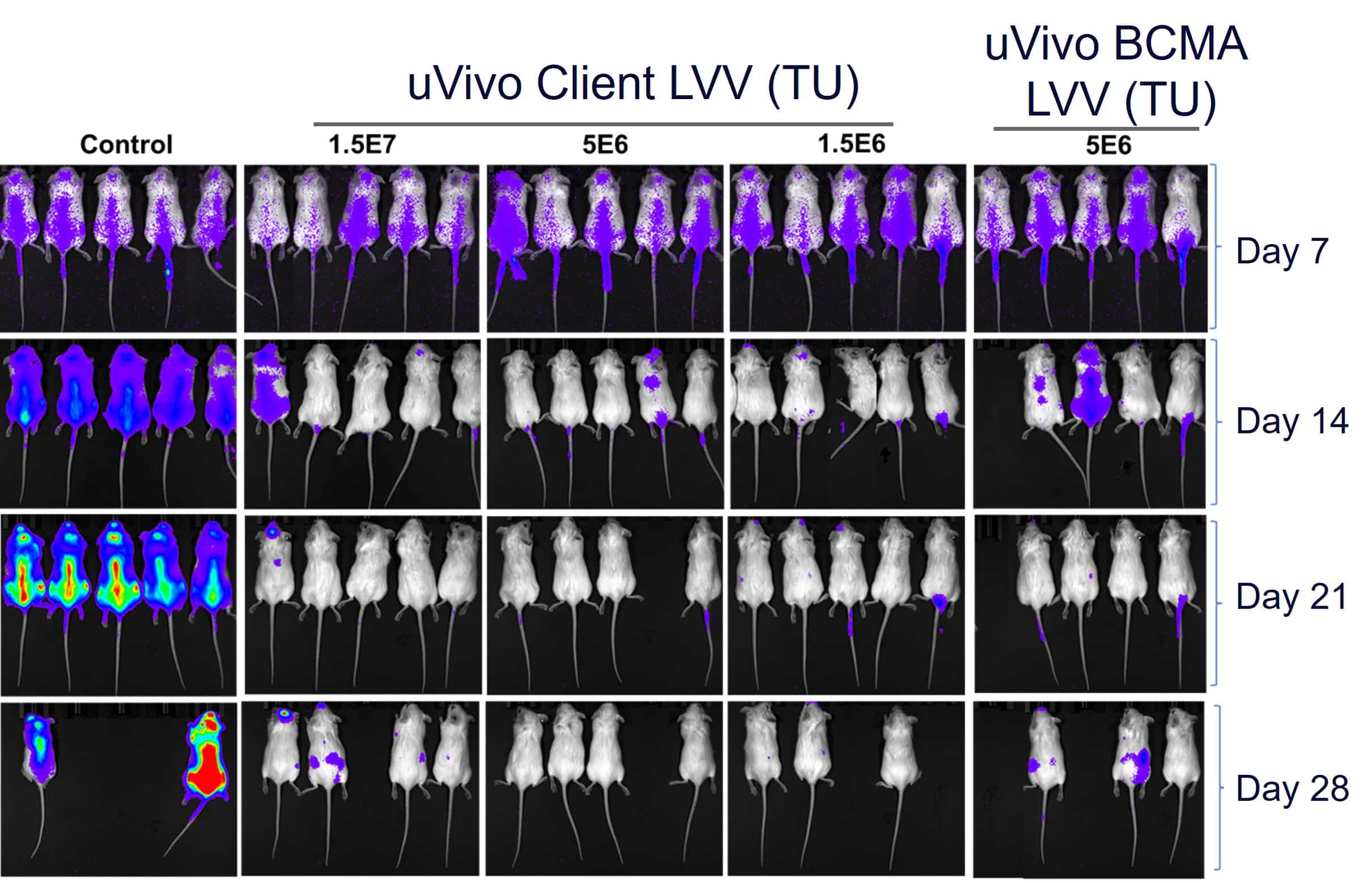
Fig. 3. uVivo CAR-T Shows Efficacy in a Multiple Myeloma Mice Model. A. Diagram of the mouse treatment schedule. NSG mice were injected intravenously (i.v.) with luciferase-labeled myeloma cells and treated with lentiviral vectors. Tumor burden was monitored by in vivo bioluminescence imaging. B. In vivo CAR LVV particles were produced using our uVivo™ LVV technology and LVV Turbo™ manufacturing platform. Three doses of the uVivo Client LVV and one dose of the uVivo anti-BCMA LVV were evaluated. Weekly bioluminescence imaging of mice to track tumor burden.
Request a quoteCAR payloads selectively target T cells (not B cells), achieving ~45% T-cell transduction in an in vitro PBMC study (Table 1).
uVivo LVV particles were not detected in non-target tissues—including the spleen, liver, brain, muscle, kidneys, heart, lung, small intestine, stomach, or bone—in a multiple myeloma biodistribution study (Table 2).
| Cell Types | Non-Treated PBMCs | uVivo LVV Treated PBMCs |
|---|---|---|
| CD3+ | 67.18% | 67.2% |
| CD19+ | 10.67% | 13.16% |
| CAR+, CD3+ | 0.34% | 44.98% |
| CAR+, CD19+ | 0.89% | 1.05% |
Table 1. uVivo™ LVV enables precise CAR delivery to T cells. PBMCs were treated with or without uVivo LVV anti-BCMC vectors. Flow cytometry was used to measure the corresponding markers.
| Mouse# | Organs | VCN (Copies/Cell) |
|---|---|---|
| BCMA085E6 | Lung | 0.0004 |
| Muscle | 0.0836 | |
| Brain | 0.0038 | |
| Small intestine | 0.0586 | |
| Spleen | 0.0019 | |
| Liver | 0.0011 | |
| Kidney | 0.0020 | |
| Heart | 0.0050 | |
| Stomach | 0.0144 | |
| Bone | 0.1424 | |
| Positive Control | 7.865 | |
| Standard Recovery Rate: 95.1% | ||
Table 2. uVivo LVV does not target other organs in the animal study. PBMCs were treated with or without uVivo LVV anti-BCMA vectors, and marker expression was assessed by flow cytometry.
Request a QuoteDiscover how our LVV Turbo platform can produce thousands of CAR-T doses.
Discover Lentivirus (LVV Turbo™) production processes and testing.
Discover CAR-T production process & regulatory guidelines.
Discover our mRNA-LNP manufacturing capabilities and testing platform
In vivo CAR-T therapies deliver CAR constructs directly into the patient—typically via lentiviral vectors or lipid nanoparticles (LNPs)—to generate CAR-T cells inside the body. CAR-T cells then engage and eradicate target cells. This approach eliminates the need for ex vivo T-cell engineering.
By simplifying the treatment process, in vivo CAR-T has the potential to accelerate therapy timelines, improve scalability, and increase accessibility and affordability for cancer patients.
In vivo CAR-T therapies are typically administered via intravenous (IV) infusion, similar to many biologic treatments. The therapeutic payload—delivered by lentiviral vectors or lipid nanoparticles (LNPs)—circulates systemically and targets T cells in the body.
In vivo CAR-T therapies typically use targeted lentiviral vectors (LVVs) or lipid nanoparticles (LNPs) to deliver CAR constructs directly into the body. Both platforms can be engineered for T-cell specificity using targeting ligands such as antibodies or nanobodies against markers including CD3, CD4, CD8, or CD7.
uBriGene’s uVivo™ LVV CAR-T platform utilizes a CD7 nanobody–based targeting strategy to selectively deliver CAR constructs to T cells, enabling efficient in vivo T-cell programming.
LVVs enable durable and persistent CAR expression via genomic integration, while LNP-based delivery is transient and may require repeat dosing.
Although VSV-G is widely used for ex vivo CAR-T applications due to its broad tropism mediated by the LDL receptor, it is susceptible to neutralizing antibodies present in human serum.
In contrast, CocalG is associated with lower levels of neutralizing antibodies and is expected to persist longer in vivo. Therefore, Cocal-pseudotyped lentiviral vectors have demonstrated resistance to inactivation by human serum.
Overall, engineered CocalG enables detargeted and retargeted LVV delivery, making it more suitable than VSV-G for targeted in vivo CAR-T applications.
In vivo CAR-T approaches deliver CAR constructs using targeted viral vectors or lipid nanoparticles, enabling T cells to be engineered directly inside the body.
In vivo CAR-T therapies use lentiviral vectors or lipid nanoparticles (LNPs) to directly program T cells inside the body after intravenous administration. This approach eliminates complex ex vivo manufacturing, reduces treatment time, and may avoid lymphodepleting chemotherapy. The same CAR vector / LNPs can be used to treat many cancer patients. As a result, in vivo CAR-T offers a faster, more scalable, and potentially more cost-effective path to cancer treatment, improving patient accessibility.
Autologous and allogeneic CAR-T therapies both rely on ex vivo T-cell engineering, while in vivo CAR-T enables direct T-cell programming inside the patient. While autologous and allogeneic approaches typically require lymphodepleting chemotherapy, in vivo CAR-T strategies may reduce or eliminate this requirement, depending on the design.
Autologous CAR-T: T cells are collected from the patient via leukapheresis, engineered and expanded ex vivo, and then reinfused. This approach is personalized but time-consuming and complex.
Allogeneic CAR-T: T cells are derived from a healthy donor and engineered ex vivo to create an off-the-shelf product. This can reduce manufacturing time but may involve risks such as immune rejection or graft-versus-host disease (GVHD).
In vivo CAR-T: CAR constructs are delivered directly into the patient (e.g., via lentiviral vectors or LNPs), enabling T cells to be engineered in situ. This approach eliminates leukapheresis and depletion conditioning, with the potential for faster, more scalable, and cost-effective therapy.
Extensive expertise with a track record of successfully releasing over 60 GMP batches of AAV.
Preclinical studies demonstrate highly specific T-cell targeting, with minimal to no transduction in non-target tissues
uVivo™ LVV is powered by detargeted Ubri-CocalG and CD7 nanobody–mediated T-cell targeting.
Research and GMP lentivirus productions
Sit aliquam elit vitae bibendum lectus blandit dictum. Nisi porta elementum sed diam eleifend pellentesque. Quis aenean quam.
Request a Quote
Optional caption section: This caption text can be used under any image or content block as-needed. Use cases are figure legends, citations, image descriptions
Ask the plasmid DNA expert! With over 300 GMP batches in our track record and our cost-effective platform technology, we can help accelerate your clinical programs.