Reliable plasmid and mRNA analytical development and QC testing services, ensuring regulatory compliance, product quality, safety, and consistency.

uBriGene offers platform-based compendial and qualified assays for plasmid and mRNA product characterization and release, adhering to the highest regulatory standards. As a leading CDMO for plasmids and mRNA, we have successfully released over 300 GMP batches.
Our skilled technical team also offers custom analytical method development, validation, and tech transfer tailored to your specific assay needs.

Optional caption section: This caption text can be used under any image or content block as-needed. Use cases are figure legends, citations, image descriptions
Reliable plasmid and mRNA-LNP AD/QC testing to ensure high quality and compliant drug products.

Optional caption section: This caption text can be used under any image or content block as-needed. Use cases are figure legends, citations, image descriptions
Recent advances in mRNA and lipid nanoparticles have expanded mRNA's role in vaccines and immuno-oncology. It is produced via in vitro transcription using plasmids as templates. Plasmid and mRNA analytical testing pose unique challenges.
The challenges in plasmid DNA quality release testing include quantifying supercoiled DNA and detecting contaminants such as endotoxins, host DNA, RNA, and proteins. mRNA quality testing requires HPLC, LC-MS, and CE to assess 5’ capping efficiency and poly(A) tail length, demanding specialized expertise and precise control.
Identity testing is crucial for ensuring the quality and safety of plasmid DNA and mRNA products used in gene therapy and vaccine production. It confirms that the final product contains the correct sequence.
For plasmids and mRNA, DNA sequencing is the primary method to determine the complete nucleotide sequence. Sequencing techniques include traditional Sanger sequencing and Next-Generation Sequencing (NGS), which enables complete sequencing without primers. Additional identity verification methods include restriction analysis.
Purity testing is fundamental in biologics manufacturing and quality evaluation, requiring reliable, robust, and accurate methods. Residual impurities are also crucial for assessing process and product quality, with sensitive methods typically developed for each impurity. The following methods are used for plasmid and mRNA products:
Plasmid and mRNA purity by UV spectrophotometer
Plasmid supercoiled percentage by HPLC
mRNA 5’ capping efficiency and 3’ polyA tail length by CE/LC-MS
mRNA fragments/integrity and aggregates by HPLC
mRNA fragments/integrity and aggregates by HPLC
dsRNA quantitation for mRNA by ELISA
Host cell DNA, RNA for plasmids by qPCR, HPLC, respectively
Host cell protein, residual protein for plasmids by ELISA, Qubit/MicroBCA, respectively
Residual DNA template for mRNA by qPCR
Residual solvents for mRNA by GC-MS
Safety testing is critical in biologics manufacturing to ensure the quality and safety of drug products.
Safety testing is critical in biologics manufacturing to ensure the quality and safety of drug products.
The recent breakthrough in lipid nanoparticle (LNP) technology has been widely used for mRNA delivery in therapeutic applications, including vaccines and cell and gene therapies. LNP analytical testing ensures quality, consistency, and delivery while complying with regulatory standards.
Identity of lipids by RP-HPLC
LNP size and polydispersity by dynamic light scattering (DLS)
Concentration with fluorescence-based assay
Purity: aggregates, encapsulation efficiency, and residual solvents testing
Safety: Sterility by culture method, endotoxin by Kinetic chromogenic LAL
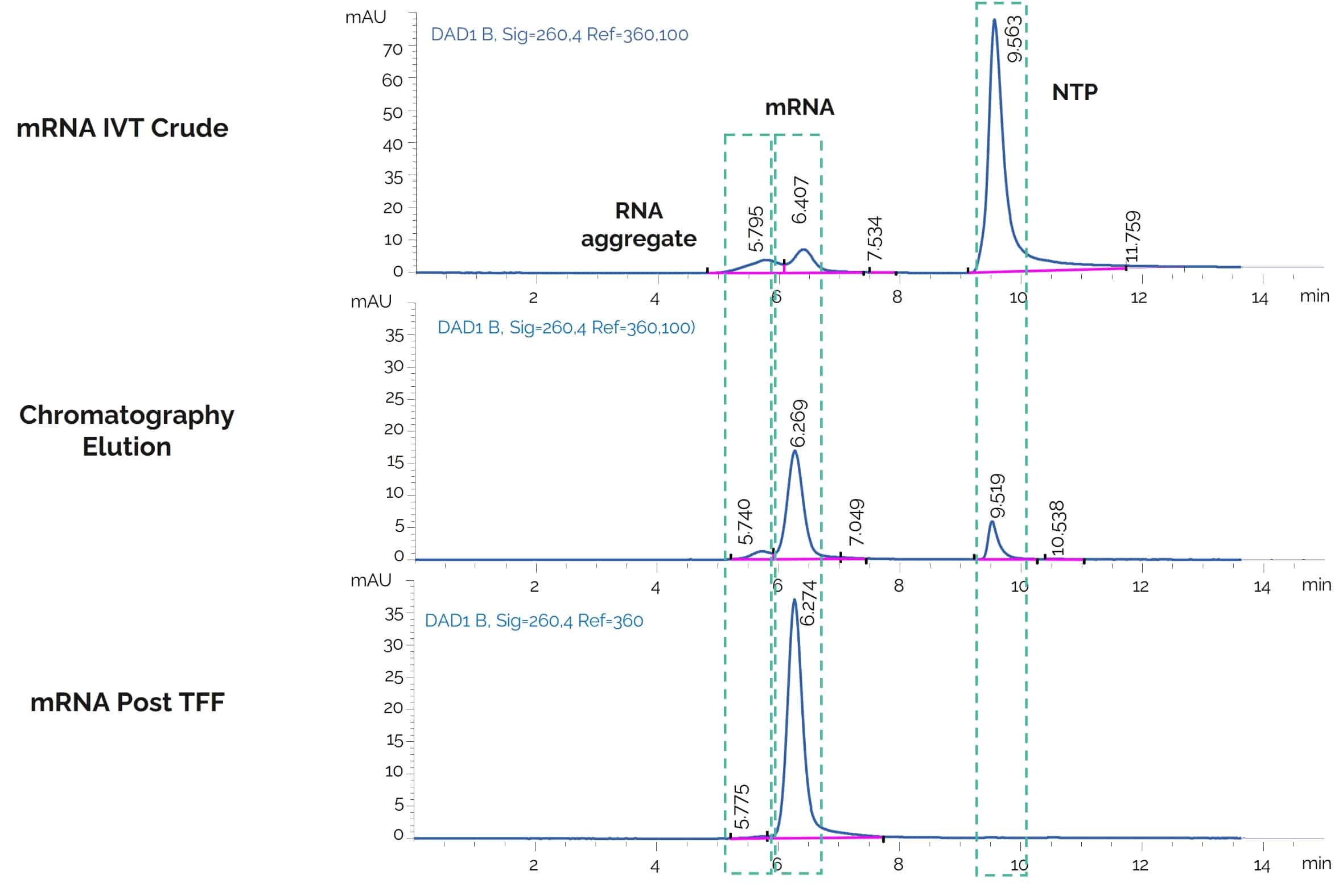
SEC HPLC analysis of mRNA post chromatography purification
Supercoiled plasmid quantitation is typically performed using HPLC (High-Performance Liquid Chromatography), which provides precise separation of different plasmid isoforms. Additional methods include agarose gel electrophoresis with densitometry analysis and capillary electrophoresis (CE) for higher resolution.
Yes, uBriGene provides standalone plasmid QC testing for plasmids manufactured elsewhere. Our comprehensive analytical services ensure regulatory compliance and product integrity, including identity, purity, potency, and safety testing.
Absolutely. uBriGene offers custom analytical development, validation, and tech transfer tailored to your specific plasmid and mRNA characterization needs. Our experts specialize in method optimization for regulatory submissions.
Yes. We provide storage/stability testing services, real-time stability and accelerated stability testing.
Key mRNA quality tests include:
1. Identity Testing – Sequence confirmation via NGS or Sanger sequencing
2. Purity Testing – 5’ capping efficiency, poly(A) tail length (HPLC, LC-MS, CE)
3. Integrity Testing – Fragmentation and aggregation assessment (HPLC, CE)
4. Safety Testing – Residual DNA, endotoxins, sterility (qPCR, ELISA, LAL assay)
Yes, uBriGene provides comprehensive mRNA-LNP analytical testing, including:
1. LNP Identity & Composition (RP-HPLC)
2. Particle Size & Polydispersity (DLS)
3. Encapsulation Efficiency & Purity
4. Sterility & Endotoxin Testing
Yes. uBriGene has extensive experience in circRNA and saRNA manufacturing, quality control release, and analytical method development.
uBriGene follows stringent FDA, EMA, NMPA, and TGA guidelines. Our GMP-compliant facility has an active DMF with the FDA, and products tested at our site have received FDA IND clearance.

Optional caption section: This caption text can be used under any image or content block as-needed. Use cases are figure legends, citations, image descriptions