High-quality exosome / extracellular vesicle products manufactured by an established exosome CDMO, ensuring consistent quality and regulatory compliance.

Mesenchymal stem cell (MSC) and iPSC-derived exosomes are offered and produced from fully characterized and released Master Cell Banks (MCBs), ensuring consistency, traceability, and quality. Purity and quality of the extracellular vesicles are enhanced by our optimized chromatography-based purification process.
Animal origin free – Safe and fully traceable
High purity – Purified using tangential flow filtration (TFF) and chromatography
Extensively tested – Comprehensive safety and potency testing performed
iPSC and MSC derived exosomes available – Manufactured from rigorously characterized mesenchymal stem cells MSCs and iPSC master cell bank
Drug Master File (DMF) for MSC derived exosomes – Supporting IND submission and regulatory review
The various Rep/Cap plasmids and helper plasmids have been tested for high quality and high yield in GMP AAV production.
We have assisted our clients in obtaining IND clearance with AAV produced using these AAV helper plasmids.
| Catalog# | Description | Particles per Vial | Product Grade | Price |
|---|---|---|---|---|
| HU-IPSC-EXOSOME | High-quality exosomes / Extracellular vesicles produced from Human iPSC master cell bank, purified via chromatography. | 20 billion particles | Research | |
| Varies | GMP | Please Inquire | ||
| HU-MSC-UC-EXOSOME | High-quality exosomes produced from the Human umbilical cord MSC bank, purified via chromatography. | 20 billion particles | Research | |
| Varies | GMP |
Note: This product is intended for in vitro research and/or manufacturing use only. Biological samples and any materials derived from these samples are for in vitro research purposes only.
Request a quoteSit aliquam elit vitae bibendum lectus blandit dictum. Nisi porta elementum sed diam eleifend pellentesque. Quis aenean quam.
Request a Quote
Optional caption section: This caption text can be used under any image or content block as-needed. Use cases are figure legends, citations, image descriptions
As an experienced exosome / extracellular vesicle CDMO, uBriGene supports preclinical and clinical studies with scalable, optimized extracellular vesicle manufacturing processes.
Extensive plasmid production experience
100+ GMP plasmid projects annually
To ensure consistency and regulatory compliance, our exosomes / extracellular vesicles are derived from iPSC and MSC master cell banks that are rigorously characterized and fully released.
The iPSC master cell bank (MCB) was generated from fibroblasts using uBriGene’s RNA-LNP reprogramming cocktail, with donor cells sourced in compliance with FDA regulations. The MCB was established using single-cell cloning technology, fully characterized, and QC released. Click to view more details of the iPSC bank.
The MSC master cell bank (MCB) was established from umbilical cord tissue sourced in compliance with FDA regulations. The GMP-grade MSC master cell banks were rigorously tested and released.
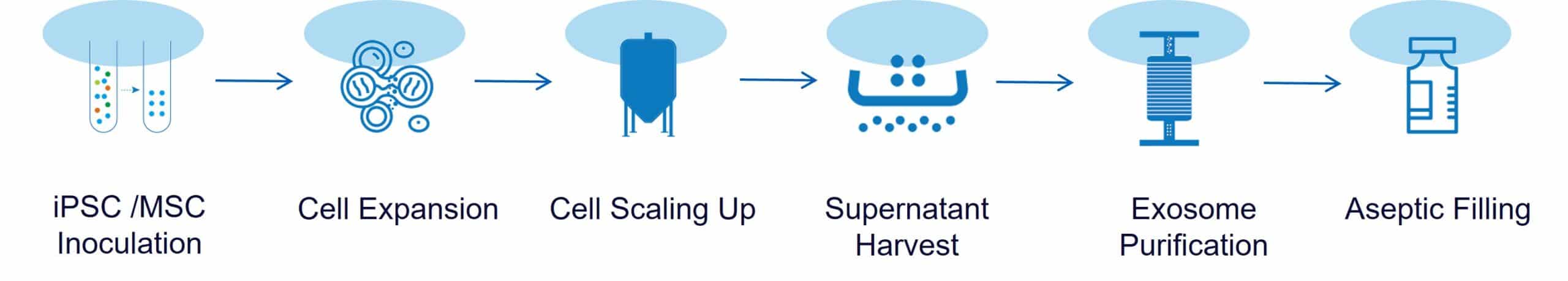
Our research-grade and GMP-grade exosomes are manufactured from extensively characterized and fully released iPSC or MSC master cell banks (MCBs). Using 2D adherent or 3D spheroid-based suspension cultures in bioreactors, we enable robust, scalable, and consistent exosome / extracellular vesicle production.
The culture supernatants are then concentrated and purified using tangential flow filtration (TFF) and chromatography to produce high-purity, high-potency exosomes / EVs.
In addition to our off-the-shelf extracellular vesicle products, we also offer custom exosome CDMO services. Contact us.
uBriGene’s lentivirus packaging plasmids were used for lentiviral vector production with uBriGene’s proprietary LVV suspension cell bank, 293TH. Transduction titer was measured using flow cytometry after transducing Jurkat cells.
HEK293 cells were transduced with GFP lentivirus. Fluorescent images taken 48 hours post transduction.
GMP-grade exosomes are rigorously tested for size distribution, protein markers, safety, adventitious viruses, and potency. The iPSC and MSC master cell banks that are used for exosome – extracellular vesicles are fully characterized and released in compliance with regulatory requirements.
| Testing | Methods | MCB | Exosome Products | |
|---|---|---|---|---|
| Sterility | Membrane filtration method | √ | √ | |
| Mycoplasma | Culture method | √ | √ | |
| Identity | STR | √ | ||
| Mycobacteria | Culture method | √ | ||
| Adventitious Viruses | In vitro cell morphology observation and hemadsorption assay | √ | ||
| In vitro assay using multiple indicator cell lines for adventitious agent detection (28-day) | √ | √ | ||
| In vivo adventitious agent testing using animal models and embryonated chicken eggs | Neonatal (suckling) mice | √ | √ | |
| Mouse Inoculation of embryonated chicken eggs via the yolk sac and allantoic cavity | √ | √ | ||
| Retrovirus examination | Reverse Transcriptase (RT) activity assay for detection of retroviral contamination (RERT) | √ | ||
| Endotoxin | Gel-clot method | √ | ||
| Physical Morphology | Transmission Electron Microscopy (TEM) | NA | √ | |
| Exosome Protein Marker | Western blot | NA | √ | |
| Exosome Particle Size | Nanoparticle Tracking Analysis (NTA) | NA | √ | |
| Potency Assays | Wound healing assay | NA | √ | |
The exosome / extracellular vesicle products have undergone rigorous QC release testing to ensure purity, safety, and potency.
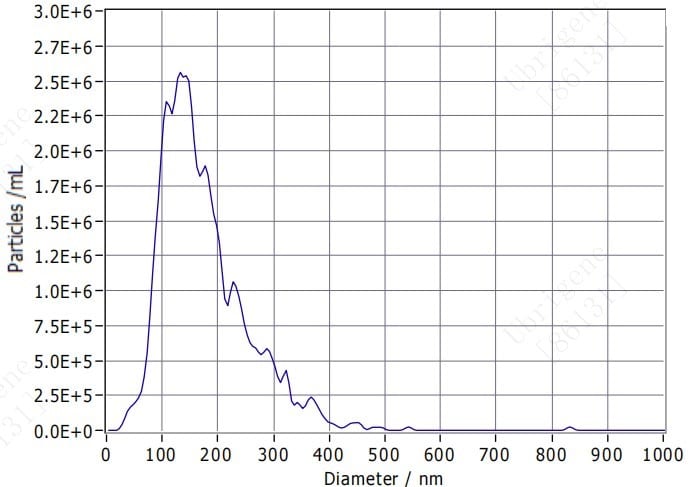
Fig. 1. NTA analysis showed that the majority of exosomes / extracellular vesicles are approximately 150 nm in diameter.
Request a quote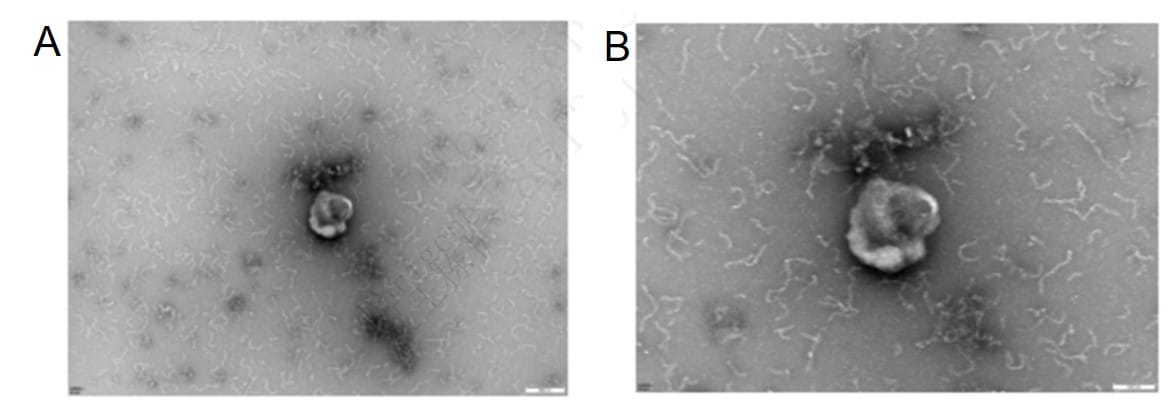
Fig. 1. NTA analysis showed that the majority of exosomes / extracellular vesicles are approximately 150 nm in diameter
Transmission electron microscopy (TEM) analysis of iPSC exosomes shows a characteristic cup-shaped morphology, with a clean background and low levels of contaminating proteins. A: 20,000x, B: 40,000x
iPSC exosomes have demonstrated wound-healing potential. A cell scratch assay using primary fibroblasts was conducted to evaluate cellular repair efficiency.

Fig. 2. iPSC exosomes were evaluated for wound-healing activity using primary fibroblasts. Both exosome / extracellular vesicle batches demonstrated greater repair activity compared to the control.
Request a quoteLearn about uBriGene’s iPSC banking and reprogramming capabilities.
Learn how to accelerate the development of iPSC-derived cell therapies.
Unlock safer regenerative therapies with Genome-Safe iPSC RPM for clinical success.
Redefining RNA Therapeutics: GMP RNA-LNP Manufacturing You Can Trust
Exosomes are extracellular vesicles (EVs), membrane-bound, tiny particles (30–150 nm) released by cells that carry proteins, lipids, and genetic material, such as mRNA, miRNA, and DNA. They play important roles in regulating biological processes and are widely studied in regenerative medicine, diagnostics, and drug delivery.
As an established exosome CDMO, uBriGene isolates exosomes / extracellular vesicles from induced pluripotent stem cell (iPSC) or mesenchymal stem cell (MSC) culture supernatants using tangential flow filtration (TFF), followed by size-exclusion chromatography to further enhance purity.
Each batch undergoes comprehensive characterization and QC release testing, including particle size distribution, particle concentration, morphology, protein markers, sterility, endotoxin, and potency assays.
Exosomes are relatively stable extracellular vesicles due to their lipid bilayer membrane. For long-term storage, exosomes are recommended to be stored at -80 °C.
Exosomes carry bioactive molecules that regulate cell communication and tissue repair. MSC derived exosomes are widely studied for their ability to promote tissue regeneration by enhancing cell proliferation and migration, stimulating angiogenesis, and modulating inflammation. Because of these properties, MSC exosomes are being explored in applications such as skin care, skin repair, chronic wound healing, musculoskeletal regeneration, and tissue engineering.
Exosomes derived from mesenchymal stem cells (MSCs) are commonly used in orthopedics due to their anti-inflammatory properties and their ability to promote chondrocyte regeneration.
Exosomes carry bioactive molecules and transfer biological materials between cells. Exosomes are commonly used in aesthetics, tissue regeneration, diagnostic biomarkers, and immunotherapies.
Exosomes derived from iPSC cell culture supernatant contain bioactive components such as proteins, lipids, and nucleic acids that have anti-aging potential. In exosome skin care products, iPSC-derived exosomes are used as cosmetic ingredients that help improve overall skin appearance by supporting skin vitality, enhancing skin smoothness, and promoting a more radiant, youthful-looking complexion.
Extensive expertise with a track record of successfully releasing over 60 GMP batches of AAV.
iPSC Cell Banks for clinical use
Research and GMP lentivirus productions
Sit aliquam elit vitae bibendum lectus blandit dictum. Nisi porta elementum sed diam eleifend pellentesque. Quis aenean quam.
Request a Quote
Optional caption section: This caption text can be used under any image or content block as-needed. Use cases are figure legends, citations, image descriptions
Ask the plasmid DNA expert! With over 300 GMP batches in our track record and our cost-effective platform technology, we can help accelerate your clinical programs.