An experienced team specializing in cGMP-compliant storage stability testing studies for cell and gene therapy products to ensure safety and quality.

As part of our CGT CDMO team, our experienced stability testing staff has designed and conducted numerous storage and stability studies for plasmids, RNA-LNP, AAV, LVV, and cell products.
Storage and stability studies are conducted in compliance with ICH guidelines, including study design, sampling, testing schedules, and requirements tailored to the specific nature of therapeutic products.

Optional caption section: This caption text can be used under any image or content block as-needed. Use cases are figure legends, citations, image descriptions

Optional caption section: This caption text can be used under any image or content block as-needed. Use cases are figure legends, citations, image descriptions
Advanced therapy products are sensitive to environmental factors, such as temperatures, including plasmids, RNA-LNP, viral vectors, and cell products.
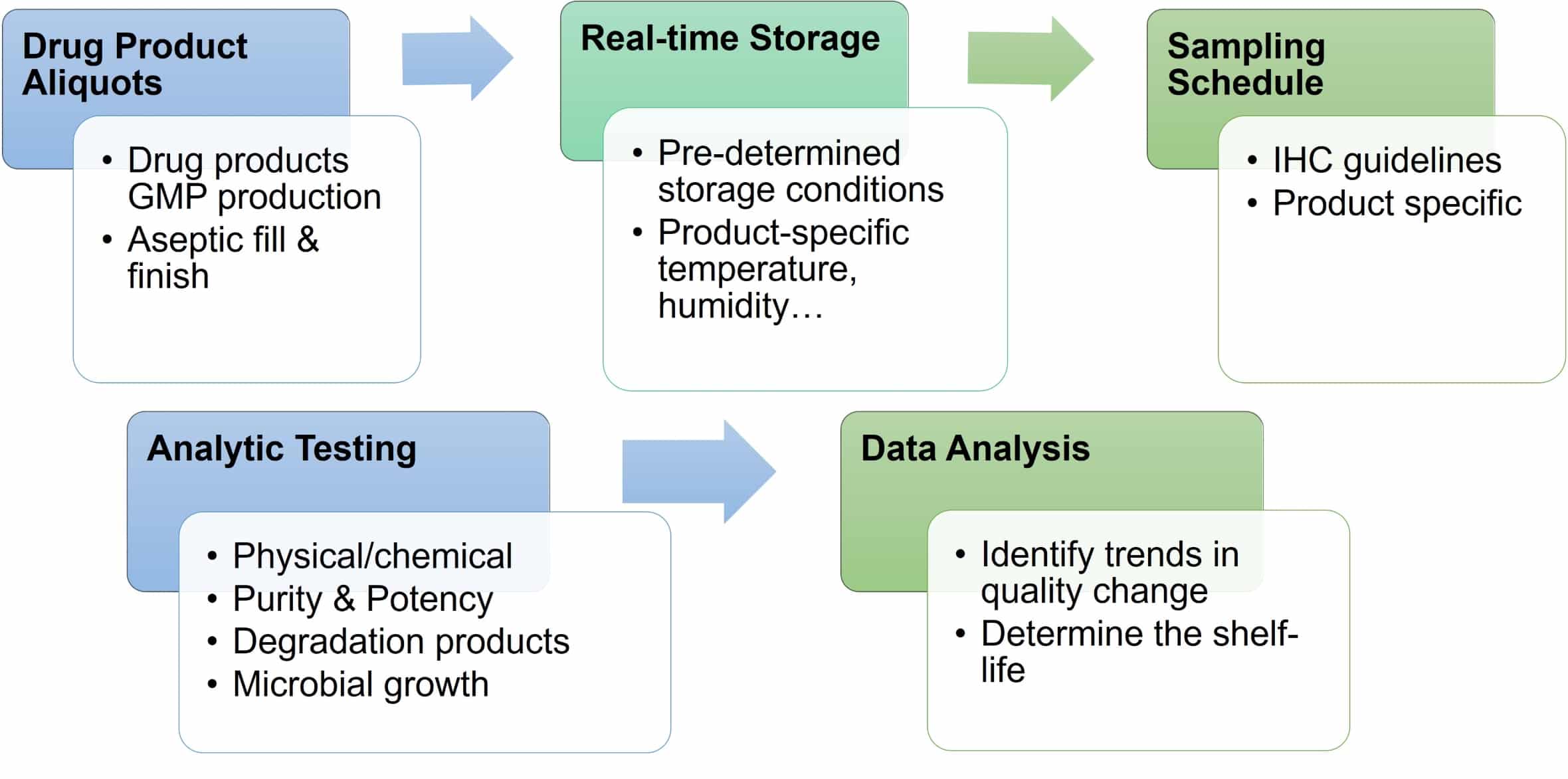
A real-time storage and stability study evaluates the quality of drug products under the intended storage conditions. A series of analytical tests will assess the chemical and physical properties, potency, and sterility at specific storage intervals in accordance with ICH guidelines.
In accelerated stability testing, biological products are exposed to elevated stress conditions, such as high temperature and/or humidity. Accelerated studies help predict product stability under normal storage conditions, providing preliminary stability data for early-stage approval processes. Additionally, these studies assist in evaluating formulation parameters for the products.
While real-time studies are essential for confirming long-term stability, accelerated studies are invaluable for expediting product development and regulatory timelines.
At defined storage time points, products undergo a series of analytical tests to evaluate their quality, including appearance, purity, structure, and biological activity.
Physiology/chemistry stability testing
Biological activity testing
Microbial testing for safety - Sterility
Learn more about our CAR-T CDMO manufacturing for high-quality therapies.
Discover Lentivirus (LVV) production processes and testing.
Explore RNA drug development and manufacturing processes.
Explore how uBriGene helped our client to receive FDA IND clearance in 9 months.
Stability testing determines how long a drug substance or formulation remains safe and effective under defined storage conditions, with evaluations conducted at specified time intervals.
Advanced therapy products, including plasmids, viral vectors, RNA, LNPs, and cell therapy products, are sensitive to environmental factors such as temperature and humidity. Storage stability testing assesses biopharmaceutical products under intended storage conditions over a specified time frame. The results help determine recommended storage and transportation conditions for drug substances and products, as well as establish appropriate shelf lives.
Real-time stability studies evaluate the quality of products under the intended storage conditions, including temperature and humidity. Samples are tested at predefined intervals to assess their quality, including appearance, purity, structure, and biological activity.
In accelerated stability testing, biological products are stored under elevated stress conditions (e.g., high temperature and/or humidity). These studies are used to predict the product’s stability under normal storage conditions and to evaluate specific formulation conditions for biological products.
Storage stability studies are conducted to evaluate the physical, chemical, and biological properties of products under intended storage conditions. The analytical testing assays may vary depending on the type of cell and gene therapy product. Stability testing typically includes assessments of appearance, color, texture, particle size, purity, potency, and sterility.
The International Conference on Harmonisation (ICH) serves as a platform for regulatory agencies worldwide to develop a unified set of guidelines for stability study requirements.
There are several established guidelines regarding the design and interpretation of stability studies. Our team conducts all stability studies in accordance with the ICH guidelines Q1A (R2): Stability Testing of New Drug Substances and Products and Q5C: Quality of Biotechnological Products – Stability Testing of Biotechnological/Biological Products.

Optional caption section: This caption text can be used under any image or content block as-needed. Use cases are figure legends, citations, image descriptions